 It contains all the geometric parameters which determine the overall shape of the molecule, as well as bond lengths, bond angles, torsional angles, and any other geometrical properties that govern the position of each atom. Molecular geometry helps in understanding the shape of the molecule by the three-dimensional arrangement of the atoms that make up a molecule. The valence-shell electron-pair repulsion (VSEPR) theory is used to determine the molecular geometry and the electron-group geometry. 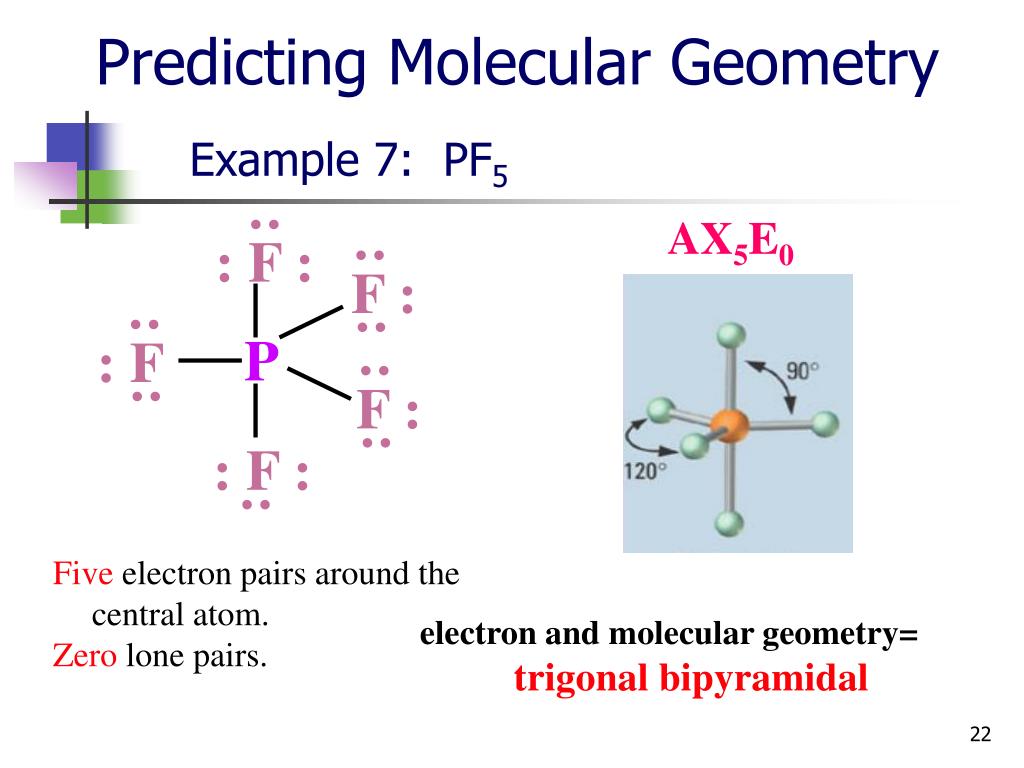
The second is to determine the electron geometry from the Lewis dot structure and lastly to determine the molecular geometry. The first is to know the Lewis dot structure of the compound which helps in to identify the bond pairs and the lone pairs. Pseudorotation is particularly notable in simple molecules such as phosphorus pentafluoride (PF 5).There are three steps that help in determining the shapes of molecules. In the process of pseudorotation, two equatorial ligands (both of which have a shorter bond length than the third) "shift" toward the molecule's axis, while the axial ligands simultaneously "shift" toward the equator, creating a constant cyclical movement. Pseudorotation is similar in concept to the movement of a conformational diastereomer, though no full revolutions are completed. Isomers with a trigonal bipyramidal geometry are able to interconvert through a process known as Berry pseudorotation. Finally, the triiodide ion ( I −ģ) is also based upon a trigonal bipyramid, but the actual molecular geometry is linear with terminal iodine atoms in the two axial positions only and the three equatorial positions occupied by lone pairs of electrons (AX 2E 3) another example of this geometry is provided by xenon difluoride, XeF 2. A T-shaped molecular geometry is found in chlorine trifluoride (ClF 3), an AX 3E 2 molecule with fluorine atoms in two axial and one equatorial position, as well as two equatorial lone pairs. 
The seesaw molecular geometry is found in sulfur tetrafluoride (SF 4) with a central sulfur atom surrounded by four fluorine atoms occupying two axial and two equatorial positions, as well as one equatorial lone pair, corresponding to an AX 4E molecule in the AXE notation. For molecules with five pairs of valence electrons including both bonding pairs and lone pairs, the electron pairs are still arranged in a trigonal bipyramid but one or more equatorial positions is not attached to a ligand atom so that the molecular geometry (for the nuclei only) is different. The VSEPR theory also predicts that substitution of a ligand at a central atom by a lone pair of valence electrons leaves the general form of the electron arrangement unchanged with the lone pair now occupying one position. Both factors decrease electron density in the bonding region near the central atom so that crowding in the axial position is less important. In general ligand apicophilicity increases with electronegativity and also with pi-electron withdrawing ability, as in the sequence Cl < F < CN. In the mixed halide PF 3Cl 2 the chlorines occupy two of the equatorial positions, indicating that fluorine has a greater apicophilicity or tendency to occupy an axial position. As examples, in PF 5 the axial P−F bond length is 158 pm and the equatorial is 152 pm, and in PCl 5 the axial and equatorial are 214 and 202 pm respectively. For molecules with five identical ligands, the axial bond lengths tend to be longer because the ligand atom cannot approach the central atom as closely. For phosphorus pentachloride as an example, the phosphorus atom shares a plane with three chlorine atoms at 120° angles to each other in equatorial positions, and two more chlorine atoms above and below the plane ( axial or apical positions).Īccording to the VSEPR theory of molecular geometry, an axial position is more crowded because an axial atom has three neighboring equatorial atoms (on the same central atom) at a 90° bond angle, whereas an equatorial atom has only two neighboring axial atoms at a 90° bond angle. 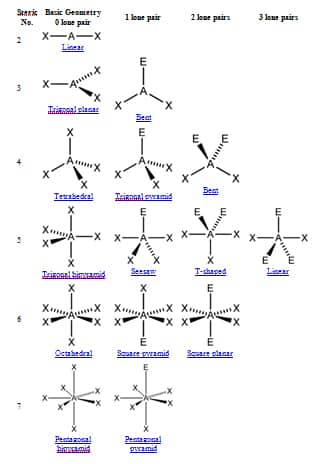
The five atoms bonded to the central atom are not all equivalent, and two different types of position are defined. Axial (or apical) and equatorial positions Trigonal bipyramidal molecular shapeĮq = equatorial ligand (in plane perpendicular to unique axis) Examples of this molecular geometry are phosphorus pentafluoride ( PF 5), and phosphorus pentachloride ( PCl 5) in the gas phase. This is one geometry for which the bond angles surrounding the central atom are not identical (see also pentagonal bipyramid), because there is no geometrical arrangement with five terminal atoms in equivalent positions. In chemistry, a trigonal bipyramid formation is a molecular geometry with one atom at the center and 5 more atoms at the corners of a triangular bipyramid. Molecular structure with atoms at the center and vertices of a triangular bipyramid Trigonal bipyramidal molecular geometry 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
